Khlood A. M. Al-Khafaji1*; Saffa A. Mahmood1; Nagam A. Moshaemash1; Mahmood A. Rhamathan1; Mohamed A. Abedalah1; Ahmed J. Feath1
1, Industrial Microbiology Department, Food and Biotechnology Centre, Agricultural Research Directorate, Ministry of Science and Technology
E-mail:
khloodalkhafaji@yahoo.com
Received: 18/09/2020
Acceptance: 12/12/2020
Available Online: 13/12/2020
Published: 01/01/2021

Manuscript link
https://dx.doi.org/10.30493/dls.2020.248860
Abstract
This research aimed to produce extracellular α-amylase from the Iraqi isolates of Bacillus thuringiensis using various agricultural materials. The initial ingredients were wheat straws, rice straws, corn cop, date seeds, reed, lathyrus seeds, corn seeds, and sorghum seeds. A colorimetric method was used to evaluate the activity of α-amylase through the determination of glucose concentration. The initial isolate investigation showed that KS4 B. thuringiensis isolate produced a significant amount of α-amylase in submerged culture. The same isolate was used in agricultural-based media experiment where 5% media of each material was prepared, inoculated with 105 bacterial spores, and incubated at 30 ○C for 48 hours. The highest α-amylase activities were obtained with corn and sorghum seeds. Later, an equilibrated environment experiment was carried out where different concentrations of corn and sorghum seeds (2.5, 5, 7.5, and 10%) were used to prepare agricultural-based broths, and each culture was incubated at either 30 ○C or 35 ○C. The results showed that 10% corn medium with 35 ○C incubation temperature resulted in the highest amylase activity.
Keywords: α-amylase, Bacillus thuringiensis, Sorghum, Corn
Introduction
Starch is one of the most abundant storage components of many economically important crops and their residues [1]. Processing starch to produce glucose through α-amylase mediated liquefaction has gained much attention lately. Despite that α-amylase enzyme is produced by a variety of living organisms, producing α-amylase from bacterial cultures has many advantages. Among these advantages are bulk production of the enzyme by bacterial biomass, ease of handling, low costs compared to other methods, different fermentation procedures to facilitate the production, the more stable produced enzyme, and no seasonal interaction [2]. The production of α-amylase enzyme has a great significance in biotechnology, constituting about 25% of the world’s total enzyme market [3]. A large number of bacterial α-amylase enzymes are commercially available and can substitute chemicals in starch and glycogen hydrolysis in starch processing industries [4]. This enzyme is an endo-amylase that releases glucose or maltose from starch and glycogen via the hydrolysis of α-D-(1-4) glucosidic bonds in the large molecules enriched with glucose unit. [5]. Besides its activity in converting starch into glucose syrup, α-amylase has a wide range of biotechnological applications in food, detergent, and pharmaceutical industries, in addition to its medicinal and analytical chemistry utilizations.
Traditionally, bacterial amylases are produced using synthetic chemical broths after adjusting their composition according to the optimal growth requirements and considering other environmental parameters. Synthetic media contents are costly, which renders producing enzymes in industrial volumes using this method uneconomical. Therefore, researchers tended to replace them with more reliable and cheaper substitutes for mass production. In this prospect, agricultural materials and their byproducts are considered a proper choice. A wide range of agricultural products and waste can be used as a carbon source in amylase production, such as millet and potato starch, wheat bran and straw, coffee waste, banana wastes, potato peel, and sugarcane bagasse [6].
Although many bacterial genera are involved in the mass production of α-amylase, spore-forming Bacillus spp are the most competent in extracellular amylases enzyme production. Previously B. cereus grown under optimal laboratory conditions to produce amylase [7], while [8] produced amylase enzyme from B. subtilis. Additionally, [9] used fruit peels as a substrate for amylase production by Bacillus sp AY3. On the other hand, B. thuringiensis is known to excrete α-amylase and many trials were conducted to produce amylases from this species [10][11]. Therefore, the main objective of the recent research was to produce α-amylase from Iraqi isolates of B. thuringiensis using agricultural-based media.
Material and Methods
Bacterial isolates
Three Iraqi bacterial isolates of B. thuringiensis were isolated from soil [12]. Two other isolates were obtained from infected dead larvae of Galleria mellonella and one final isolate from infected Ephestia kuehniella larvae. Infected larvae were crushed with sterilized distilled water. Then, the suspension was heat-treated at 80 ○C for 30 min. A loopful of suspension was streaked over nutrient agar and incubated at 28 ○C for 72 h. Isolated bacteria were identified according to [12]. The six bacterial isolates of B. thuringiensis were refreshed from their stocks using nutrient agar plates. Cultures characteristics and spore formation were examined under microscope.
Nutrient agar based medium augmented with 1% starch was prepared and sterilized at 121 ○C for 15 minutes. Each bacterial isolate was streaked over starch medium and incubated at 30oC for 72 h. Later, iodine solution was added to the growth and left for 10 minutes. Clear zones developed around the colonies were measured in response to bacterial growth [13].
Production of α-amylase in broth culture
B. thuringiensis isolates were grown in nutrient broth and nutrient broth augmented with 1% starch (analytical grade). broths were incubated for 48 h at 30 ○C in a shaker incubator (150 rpm) [14]. bacterial cultures were centrifuged at 10000 rpm for 10 min, and the supernatant bacterial cell extracts were collected and used in the quantification of α-amylase enzyme activity [11].
α-amylase activity was measured using starch as a substrate and 3,5-dinitrosalicylic (DNS) as an indicator for sugar reduction. DNS reagent was prepared by dissolving 1g of DNS in 20 ml of 2M of NaOH, then 30 g of sodium potassium tartrate was added slowly to the mixture. The DNS solution volume was completed to 100 ml with distilled water and warmed to 50 ○C until complete dissolution. 3,5-dinitrosalicylic solution was stored in a dark bottle until use [15]. The substrate solution composed of starch 1% was suspended in 0.2 M phosphate buffer (pH=7). 0.2 ml of free cell extract was mixed well with 0.8 ml of substrate solution and incubated at 30 ○C for 30 min. Then, 1.5 ml of DNS solution was added to the mixture and incubated at 100 ○C for 10 min. A red color was developed in response to glucose concentration in the solution. Red color intensity was measured using a spectrophotometer at a wavelength of 540 nm. One α-amylase activity unit was defined as the amount of enzyme which released 1 μmol of maltose or glucose equivalent per min under the assay conditions [15][16]. Regarding the enzyme activity, B. thuringiensis isolate KS4 was chosen for further experimentation.
Agricultural-based media
Agricultural components (Wheat straws, Rice straws, Corn cops, Date seeds, Reed, Lathyrus seeds, Corn seeds, and Sorghum seeds) were all obtained from Iraqi agricultural sources. Each material was ground to coarse particles and sieved out through a 2 mm mesh. 5 g of each agricultural component was added separately to 100 ml distilled water, and the pH of each medium was adjusted to 7.2±0.2. Ready to use nutrient broth (Oxoid) was prepared as described by the manufacturer and augmented with 1g of starch (1%). All media were autoclaved for 20 minutes at 121 ○C and 1.5 bar and then were cooled to 25oC.
The spore suspension was prepared from 72 h old culture, and spores were counted using a haemocytometer. Serial dilutions were prepared, and 0.1 ml of each culture was spread over nutrient agar to estimate the viable bacterial count using the equation:
Colonies forming unit/ml = Number of colonies × Reverse dilution factor × 10
Agricultural-based broths and the 1% starch-nutrient broth were inoculated with 105 spores, based on the previous estimation. The broths were incubated for 48h at 30 ○C in a shaker incubator (120 rpm). α-amylase was extracted, and its activity was estimated, as mentioned above.
After that, different concentrations of corn and sorghum seed (2.5, 5, 7.5, and 10%) were used to prepare agricultural-based broths, and each culture was incubated at either 30 ○C or 35 ○C to assess the best environmental requirements.
Statistical analysis
Statistical analysis was done using the 12th edition of gene stat. Three replications of each treatment were subjected to LSD test at significance level of 5%.
Results and Discussion
Bacterial isolate α-amylase production
Clear zones developed after iodine treatment indicated that different B. thuringiensis isolates produced different quantities of amylase at the solid medium. Most Bacillus species, including B. thuringiensis, are able to excrete α-amylase in a starch-containing medium [17]. [16] confirmed the production of amylase enzyme from many soil Bacillus isolates through the use of iodine on starch agar medium, and the developing of a clear zone around bacterial growth indicated for amylase activity.
Results showed that amylases production significantly increased in nutrient broth augmented by 1% starch compared to starch-free media (Table 1). This result is in accordance with other researches which showed that starch at concentrations of 1-2% combined with organic nitrogen-based compounds such as peptone, triptone, beef extract or yeast extract was very promising in amylase production from many Bacillus spp [18-20]. The highest amylase activities were observed in Bt KS4 (an Iraqi soil isolate) followed Bt 5G and Bt 3G, which were isolated from the infected Galleria mellonella larvae (Table 1).
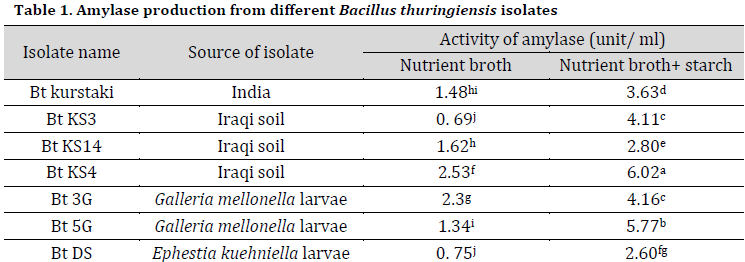
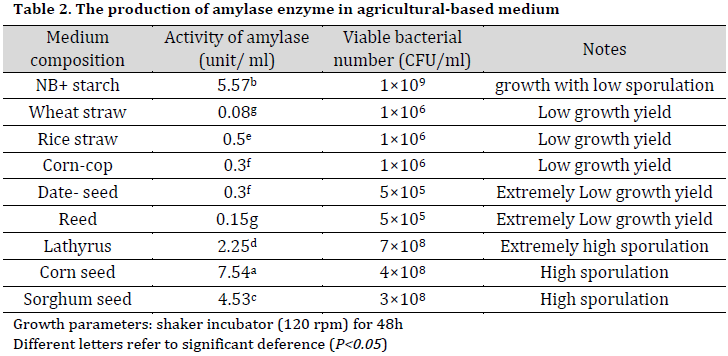
α-amylase production in agricultural-based media
Based on its performance in the initial tests, Bt KS4 was chosen for the later agricultural-based media tests. The results revealed that corn seeds medium gave the highest enzyme activity results among the tested media, including starch supplemented broth (Table 2). On the other hand, the best sporulation of Bt KS4 isolate was observed using Lathyrus based medium followed by sorghum and corn seeds. Many factors might interact with the sporulation process in B. thuringiensis, including environmental parameters and nutrients that sustain bacterial growth. The high sporulation in lathyrus based broth might be due to the chemical composition of Lathyrus seed, which contains proteins, a low level of reducing sugar, and many elemental minerals such as magnesium, phosphorous, calcium. Quorum sensing signals secreted by bacterial cells in response to cell density, nutrients, and environment are responsible for the division, sporulation, and biofilm formation through a phosphorylation regulatory system. This system demands excessive amounts of phosphorous, and Lathyrus seeds can supplement the medium with phosphorus needed to sustain this system [21-23].
Low bacterial growth was observed in wheat straw, rice straw, corn cop, and date seed media. This finding might be due to the biochemical composition of these agricultural wastes, which is rich in cellulose (30-45%), hemicellulose (20-25%), and lignin (15-20%) with a minor amount of sugars and proteins [24].
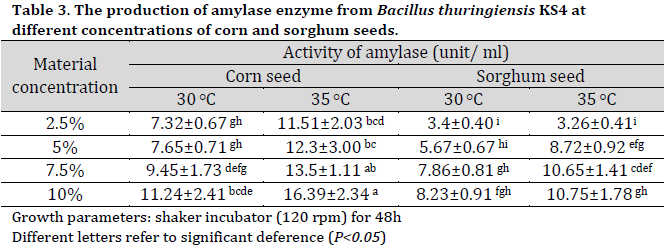
Medium optimization experiment results
Based on the superior performance of corn seed and sorghum seed media, they were chosen for further investigation with Bt KS4 isolate under different material concentration in the medium (2.5, 5, 7.5, and 10%) and two different incubation temperatures (30 and 35 °C). The highest amylase production was observed at 10% corn seed medium at 35°C with an amylase activity of 16.39 (Table 3). The main advance in the recent research was the production of α-amylase enzyme from B. thuringiensis isolated from Iraqi soil (KS4) using agricultural-based culture media of corn or sorghum seed without the need for any extra additives to fermentation media. The current results are mainly due to corn seeds’ high carbohydrates (up to 80.62%) and protein (up to 10.95%) contents as well their essential minerals and vitamins [25]. Additionally, the main carbohydrate source in corn seed and sorghum seed is starch with 59.74% and 60.02%, respectively [26].
The current results were in accordance with other successful researches that aimed to produce α-amylase using agricultural-based media for fermentation, such as Tapioca liquid waste [27], banana wastes [3], and fruit peels [9].
References
| 1 | Pfister B, Zeeman S C. Formation of starch in plant cells. Cell Mol. Life Sci. 2006;37: 2781- 807. DOI |
| 2 | Aiyer PV. Amylases and their applications. Afr. J. Biotechnol. 2005; 4(13):1525-9. |
| 3 | Unakal C, Kallur RI, Kaliwal BB. Production of α-amylase using banana waste by Bacillus subtilis under solid state fermentation. Eur J Exp Biol. 2012;2:1044-52. |
| 4 | Van der Maarel MJ, Van der Veen B, Uitdehaag JC, Leemhuis H, Dijkhuizen L. Properties and applications of starch-converting enzymes of the α-amylase family. Journal of biotechnology. 2002;94(2):137-55. DOI |
| 5 | Pandey A, Nigam P, Soccol CR, Soccol VT, Singh D, Mohan R. Advances in microbial amylases. Biotechnol. Appl. Biochem. 2000;31(2):135-52. DOI |
| 6 | Saxena R, Singh R. Amylase production by solid-state fermentation of agro-industrial wastes using Bacillus sp. Braz. J. Microbiol. 2011;42(4):1334-42. DOI |
| 7 | Halder D, Biswas E, Basu M. Amylase production by Bacillus cereus strain BRSC-S-A26MB under optimal laboratory condition. Int. J. Curr. Microbiol. App. Sci. 2014;3(6):1035-47. |
| 8 | Oyeleke SB, Oduwole AA. Production of amylase by bacteria isolated from a cassava waste dumpsite in Minna, Niger State, Nigeria. Afr. J. Microbiol. Res. 2009;3(4):143-6. DOI |
| 9 | Siddiqui A, Salahuddin T, Riaz A. Production of amylase from Bacillus sp. AY3 using fruit peels as substrate. FUUAST j. biol. 2014;4(2):213-5. |
| 10 | Chattopadhyay P, Banerjee G, Mukherjee S. Recent trends of modern bacterial insecticides for pest control practice in integrated crop management system. 3 Biotech. 2017;7(1):60. DOI |
| 11 | Smitha RB, Jisha VN, Sajith S, Benjamin S. Dual production of amylase and δ-endotoxin by Bacillus thuringiensis subsp. kurstaki during biphasic fermentation. Microbiology. 2013;82(6):794-800. DOI |
| 12 | Alkhafaji KA, Khleoy SO, Khamis HM, AbidAlrahem MS, AbidAlhadi SS. Isolation and identification of bacterium Bacillus thuringiensis from Iraqi soil and it’s uses against larvae of Ephestia cautella (Welker). J. Biotech Res. cent. 2017;11(2). |
| 13 | Madigan MT, Clark DP, Stahl D, Martinko JM. Brock biology of microorganisms 13th edition. Benjamin Cummings. 2010. |
| 14 | Luang-In V, Yotchaisarn M, Saengha W, Udomwong P, Deeseenthum S, Maneewan K. Isolation and Identification of Amylase-producing Bacteria from Soil in Nasinuan Community Forest, Maha Sarakham, Thailand. Biomed. Pharmacol. J. 2019;12(3):1061-8. DOI |
| 15 | Miller GL. Use of dinitrosalicylic acid reagent for determination of reducing sugar. Anal. Chem. 1959;31(3):426-8. DOI |
| 16 | Singh RN, Bahuguna A, Chauhan P, Sharma VK, Kaur S, Singh SK, Khan A. Production, purification and characterization of thermostable α-amylase from soil isolate Bacillus sp. strain B-10. J. Biosci. Biotechnol.. 2016;5(1):37-43. |
| 17 | Benjamin S, Smitha RB, Jisha VN, Pradeep S, Sajith S, Sreedevi S, Priji P, Unni KN, Josh MS. A monograph on amylases from Bacillus spp. Adv Biosci Biotechnol. 2013;4:227-41. DOI |
| 18 | Narang S, Satyanarayana T. Thermostable α‐amylase production by an extreme thermophile Bacillus thermooleovorans. Lett. Appl. Microbiol. 2001;32(1):31-5. DOI |
| 19 | Qader SA, Bano S, Aman A, Syed N, Azhar A. Enhanced production and extracellular activity of commercially important amylolytic enzyme by a newly isolated strain of Bacillus sp. AS-1. Turk. J. Biochem. 2006;31(3):135-40. |
| 20 | Srivastava RA, Baruah JN. Culture conditions for production of thermostable amylase by Bacillus stearothermophilus. Appl. Environ. Microbiol. 1986;52(1):179-84. |
| 21 | Chavan UD, Shahidi F, Bal AK, McKenzie DB. Physico-chemical properties and nutrient composition of beach pea (Lathyrus maritimus L.). Food Chem. 1999;66(1):43-50. DOI |
| 22 | Arslan Y, Trak D, Yildirim B, Genç H, Kendüzler E. Chemical Composition of Seeds of Two Endemic Lathyrus L. Species from Turkey. J. Biol. Life Sci. 2018;9(2):66-77. |
| 23 | Bressuire-Isoard C, Broussolle V, Carlin F. Sporulation environment influences spore properties in Bacillus: evidence and insights on underlying molecular and physiological mechanisms. FEMS Microbiol. Rev. 2018;42(5):614-26. DOI |
| 24 | Antongiovanni M, Sargentini C. Variability in chemical composition of straws. Options Mediterraneennes Serie Seminaires. 1991;16:49-53. |
| 25 | Sindhu CS, Grewal RB. Physical and chemical properties of corn varieties (HQPM-1 and HQPM-7). Int. J. Chem. Stud. 2018;6(3):3380-2. |
| 26 | Puga AP, de Mello Prado R, Mattiuz BH, do Vale DW, Fonseca IM. Chemical composition of corn and sorghum grains cultivated in Oxisol according to different application methods and doses of zinc. Cien. Inv. Agr. 40 (1): 97-108. Int. J. Agric. Nat. Resour. 2013;40(1):97-108. |
| 27 | Zusfahair Z, Ningsih DR, Kartika D, Fatoni A. Amylase from bacillus thuringiensis isolated from tapioca waste: isolation, partial purification and characterization. Mal. J. Fund. Appl. Sci. 2016;12(1):22-7. DOI |
Cite this article:
Al- Khafaji, K., Mahmood, S., Moshaemash, N., Rhamathan, M., Abedalah, M., Feath, A. The production of α-amylase enzyme from Iraqi isolate of Bacillus thuringiensis using agricultural-based media. DYSONA – Life Science, 2020;2(1): 6-11. doi: 10.30493/dls.2020.248860
